
This page is preliminary and under construction.
Pteropods are small gastropod molluscs. They are distinguished,
at an intermediate stage of development,
by the presence
of both ciliary bands, and a pair of small wings, both
of which are used in swimming. Since the swimming is at an
intermediate Reynolds number of order 50, these pteropods
find themselves swimming above
the Stokesian realm, where
cilia predominate, and below the Eulerian realm, where
wings tend to dominate. An understanding of how both are
used in different ways during the development of the
pteropod may help us understand something about the emergence
of winged flight.
We are dealing with two species of pterobods. Clione
Limacina, is a shell-less
pteropod, shown here swimming from right to left.
The longest body length we have seen so far is about 15mm,
although lengths up to 85 mm
have been reported in subarctic Atlantic regions. Here
is a view of a mature Clione:

The second species is Limacina helicina, a shelled mollusc:
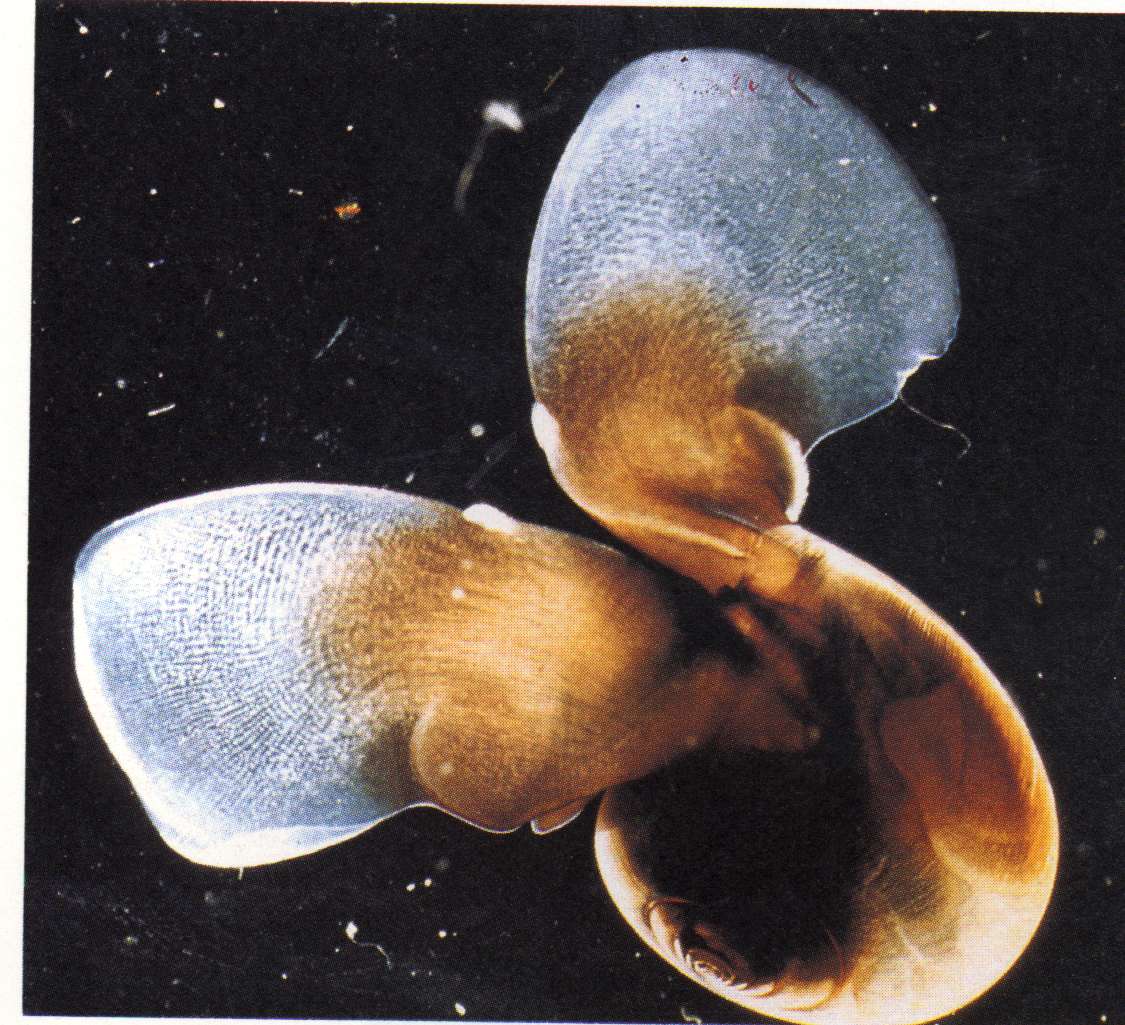
Limacina is the exclusive prey item for Clione,
and a specialized
feeding apparatus is used to extract the body from the
shell..
Clione, on the other hand are not palatable to
fish.
A basic reference: Pelagic Snails by Carol M. Lalli
and Ronald W. Gilmer, Stanford University
Press, 1989.
Experimental goals and procedures
We have collected pteropods either by directly sampling
of surface water or (usually) by drop tows
of a plankton net, through holes in the ice or at the
ice edge. The creatures may be kept in cold
sea water for up to five or six days, after which we
sacrifice them in an ethanol solution for later
chemical analysis. The main experiment is in a small
(10 cm diameter) cylindrical beaker in a constant
temperature bath. A mirror at 45 degrees allows an overhead
digital video camera to get both top and side
views simultaneusly. A cone of filter paper set into
the bottom of the beaker helps guide a descending pteropod
to a position in the field of view. Video clips
are recorded for three different modes: flapping, ciliary
swimming, and passive drifting. The flapping "flight"
is performed by a pair of small wings. At the
stage of development we are seeing now, pteropods have
three ciliary bands at head, waist, and tail, together with
the wings. The drifting is due to buoyancy, which may
be positive or negative, with neither propulsive mode active.
We seek three examples of each type of movement, for three
temperatures, -2,0,+2 degrees Centigrade,
and in sea water 0, 1, and 2% concentration of Dextran
by weight, or 81 observations per individual. (Dextran is a carbohydrate
of high average molecular weight. It does not have any apparent effect
on the pteropods,
because the osmotic effects are negligible (on the basis
of other experiments).
However it does alter the fluid density, and this can
change the buoyancy of the organism. In fact Pteropods
are seen to have control over their buoyancy,
by a process not well understood, and their density is
sufficiently close to water that the Dextran
can change te sign of buoyancy, in Clione at any
rate.
Since the viscosity of sea water increases with decreasing
temperature, our protocol decreases viscosity
in two steps by temperature, and increases it in two
steps by Dextran. We can thus separate out effects of
temperature and viscosity on motility.
Here is a view of the experimental beaker in the bath:
Robert Dudley is at the controls:
We also have an independent setup, shown below,
where analog and digital
video may be taken through a dissecting microscope. The
enables
casual observation of the behaviors, and the clips will
be used to quantify the flapping movements.
The analysis of the data, which at this writing (December
8) is only beginning, will yield
among other quantities the speed of the path projected
onto the plane of the lateral view,
and the frequency of the wing beat in the flapping
mode. We will also analyse the
wing beat pattern and try to determine a standard configuration
of the wings over a cycle.